
COMPOUNDED SEMAGLUTIDE ORAL LIQUID FOR UNDER THE TONGUE ADMINISTRATION

- Daily Dose & Easily
- Adjustable
- Compliant & No Supply Issues
- No Shots. No Needles.
- No Fridge.
- Smoother
- Drug Conc. Curves*
- Drug Delivery Study**
* Taking smaller doses more frequently (once daily) can reduce the difference between trough and peak drug levels, leading to a smoother titration and a smoother steady state concentration as compared to once weekly dosing.
** A recent in vitro study demonstrated the ability of our compounding base to deliver semaglutide into and through human gingival and oral tissues.
Compounded Semaglutide Oral Suspension for Sublingual Administration
Page Navigation Links
GLP-1 Receptor Agonists
The usage of GLP-1 receptor agonists has grown worldwide. This class includes several different injectable medications, as well as an orally delivered drug. Many patients would prefer to avoid injections if possible, and there is an extremely low absorption of oral tablets (less than 1% per the labeling for RYBELSUS®).
For these reasons and other potential valid reasons, prescribers and patients may prefer a patent-pending compounded formulation of semaglutide.
What is Sublingual Semaglutide and How is it Different?
Sublingual (SL) Semaglutide is a compounded suspension of semaglutide that is designed to be administered under the tongue (sublingually). Our patent-pending formulation utilizes a special base designed with the intent to carry large molecules (high molecular weight substances), such as semaglutide, through the oral mucosal membrane. This self emulsifying base has permeation-enhancing and unique mucoadhesive properties that help drive molecules into mucosal tissue.
A recent in vitro study demonstrated the ability of our compounding base to deliver semaglutide into and through human gingival and oral tissues.
What is the Dosage and How is it Supplied?
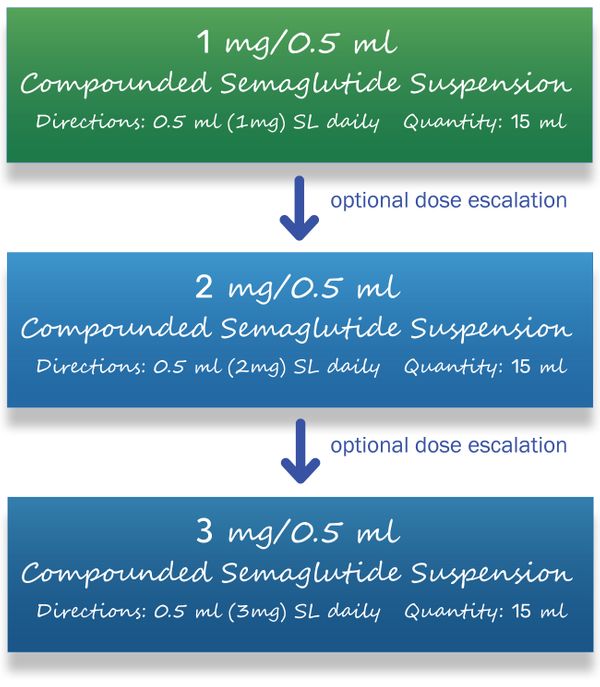
Providers:
An important consideration when titrating the dose is that semaglutide has a half-life of approximately 7 days and will usually take 4-5 weeks to reach steady state drug concentrations. Also, since the doses are daily instead of weekly, patients may experience a smoother titration and smoother steady state concentration.
Patients:
If you are starting semaglutide for the first time, you may be instructed to begin with a titration as directed by your provider. This means that you will start with a smaller dose and increase over time. Titrating this medication can help reduce gastrointestinal side effects and nausea. After a dosage increase it can take up to 4-5 weeks to reach maximum medication levels for that dose.
The medication comes in an amber bottle with a 1ml dropper for easy under-the-tongue administration. It should be stored at room temperature and shaken before use. The dose determined by your doctor should be applied under your tongue for at least 5 minutes and then swallowed. Avoid eating or drinking anything for at least 30 minutes after administration.
How much does it Cost?

How do I Get (Or Prescribe) Sublingual Semaglutide?
Providers:
We Accept:
- Faxed Rx's to 732-381-7665 (see the below fax order form)
- Phoned in Rx's to 732-381-7860
- E-scribed Rx's to Colonia Pharmacy by (1) selecting Rybelsus® 14mg tabs and (2) type the following into the sig field: semaglutide 1mg/0.5ml OR 2mg/0.5ml OR 3mg/0.5ml, 0.5ml daily, quantity: 15ml
Patients:
Sublingual Semaglutide is available with a valid prescription from your healthcare provider. Once we have a prescription we will reach out to you with more details.
We created a prescription order form (see below) that you may print and bring to your appointment to discuss this medication with your provider. (The second page of this document will provide more information for your provider) Our Fax # is 732-381-7665.
Additional Links and White Papers:
The links below are being worked on and will house a great deal of information related to this patent-pending compounded formulation as well as white papers related to Semaglutide and GLP-1 receptor agonist medications in general.
What is Semaglutide?
Semaglutide is a GLP-1 receptor agonist. GLP-1 receptor agonists usage has grown worldwide. This class includes several different injectable medications, as well as an orally delivered drug.
Semaglutide has been used for years to help patients with type 2 diabetes control their blood sugar. It has several different indications for patients with diabetes, in which it is marketed by Novo Nordisk under the brand names Ozempic®, an injectable, and Rybelsus®, an oral tablet. In 2021 the FDA approved another semaglutide containing injectable drug, Wegovy®, to help patients with weight management. These medications have been described by many to suppress one’s appetite and reduce food cravings. A suppressed appetite and reduced food cravings can lead to a decrease in caloric intake.
Although there are specific FDA-approved indications that are available from reviewing the Prescriber Information (“PI”) included in the labeling of the respective medications, you can often look at past or ongoing clinical trials for information about the medication. Please see the Clinical Trials white paper above for more information.
Compounded Sublingual Semaglutide Suspension is a compounded medication, and the FDA does not review compounded medication for safety or efficacy.
What are the Side Effects?
The most common side effects include gastrointestinal related symptoms such as abdominal pain, constipation, diarrhea, nausea, and vomiting. These side effects may be more common after initiation and during dose escalation. Extreme sensitivity reactions such as rash, trouble breathing, swelling of the mouth, face, or throat will require immediate medical attention. Low blood sugar can occur, especially if taking medication for diabetes. If a low blood sugar is suspected, discontinuing the use of the medication may be necessary should these reactions occur.
The FDA does not review any compounded medication from any pharmacy for safety or efficacy. RYBELSUS®, WEGOVY®, and OZEMPIC® are registered trademarks of Novo Nordisk A/S. Formulas and/or material listed are not to be interpreted as a promise, guarantee or claim of therapeutic efficacy or safety. The information contained herein is not intended to replace or substitute for conventional medical care, or encourage its abandonment. Every patient is unique, and formulas should be adjusted to meet their individual needs.




